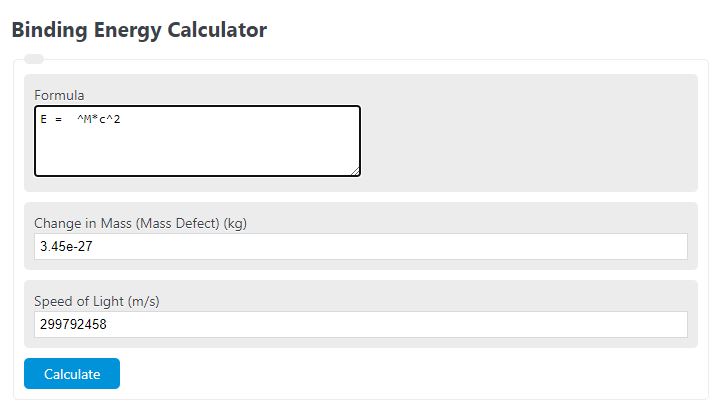
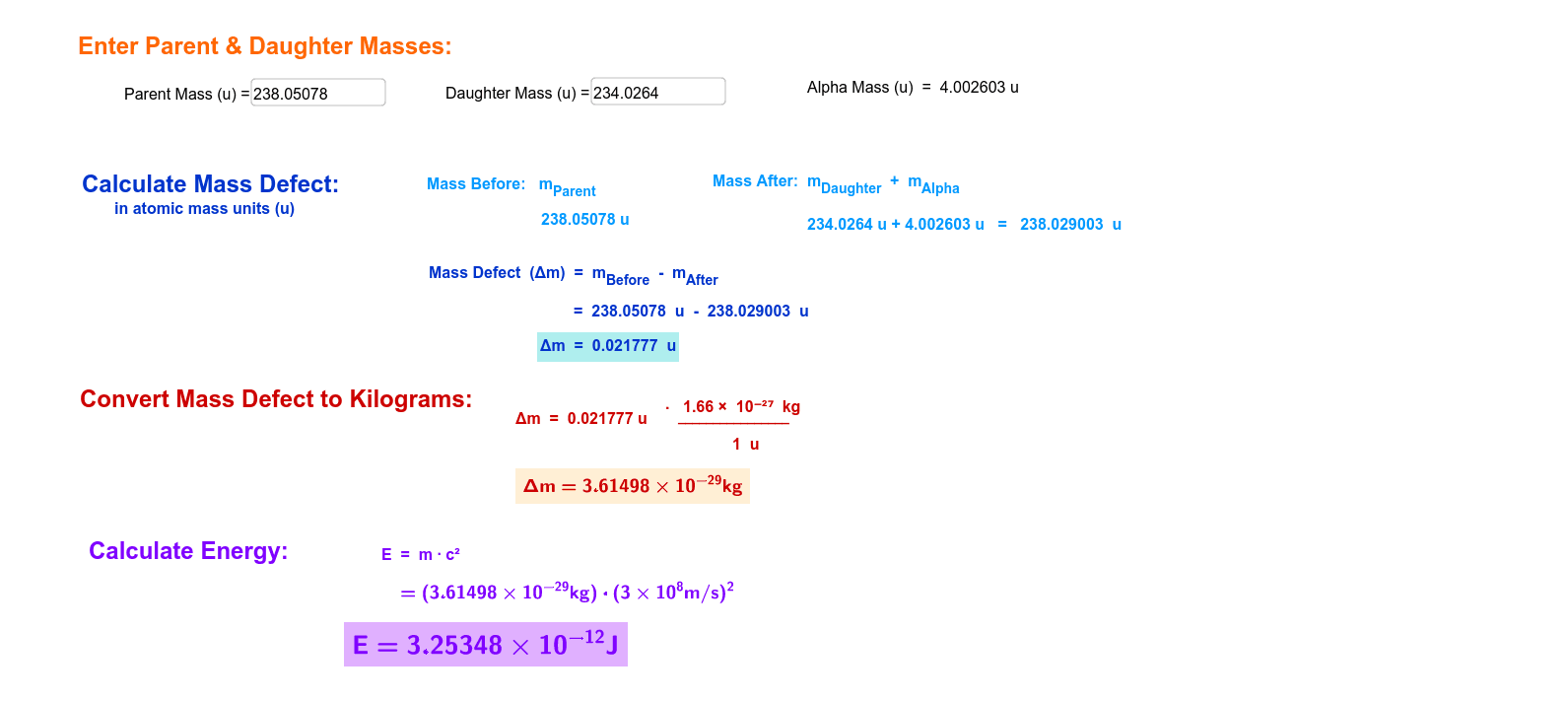
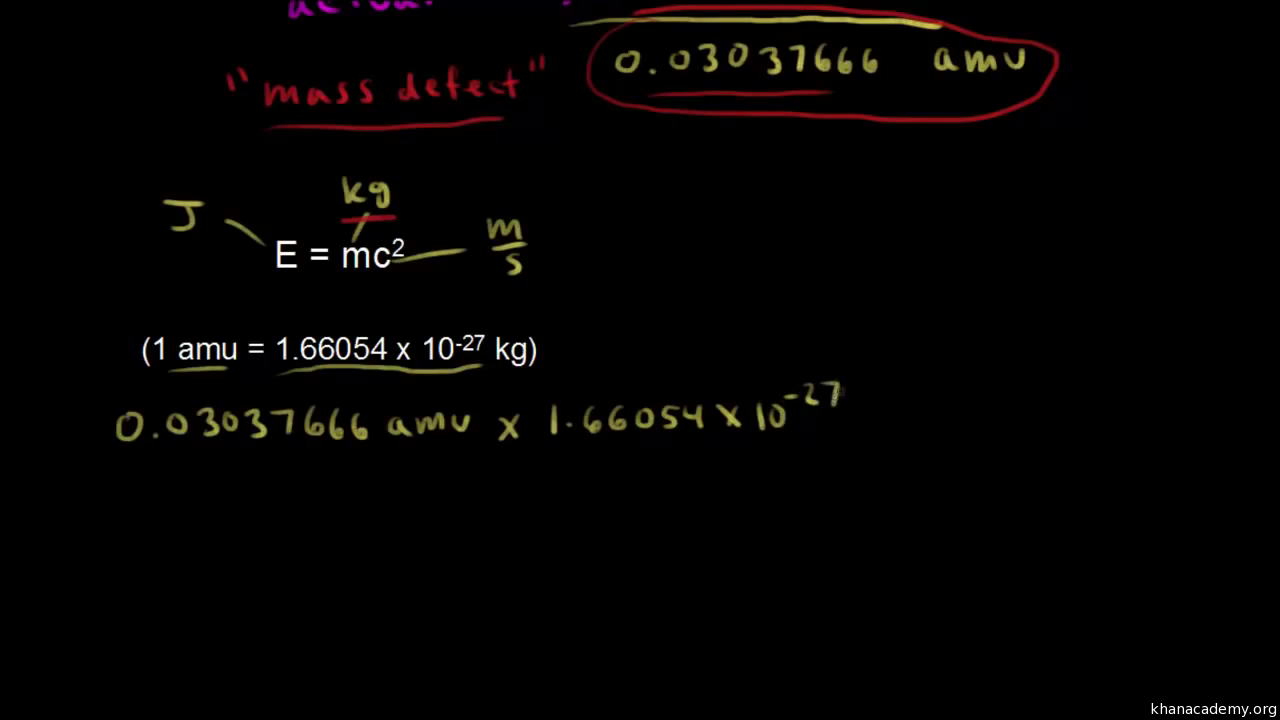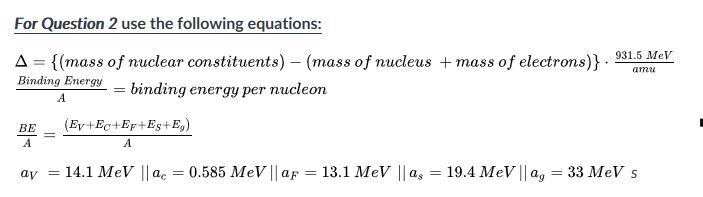Calculate the (i) mass defect, (ii) binding energy and (iii) the binding energy per nucleon for a 6C^12 nucleus. Nuclear mass of 6C^12 = 12.000000 a.m.u., mass of hydrogen nucleus = 1.007825
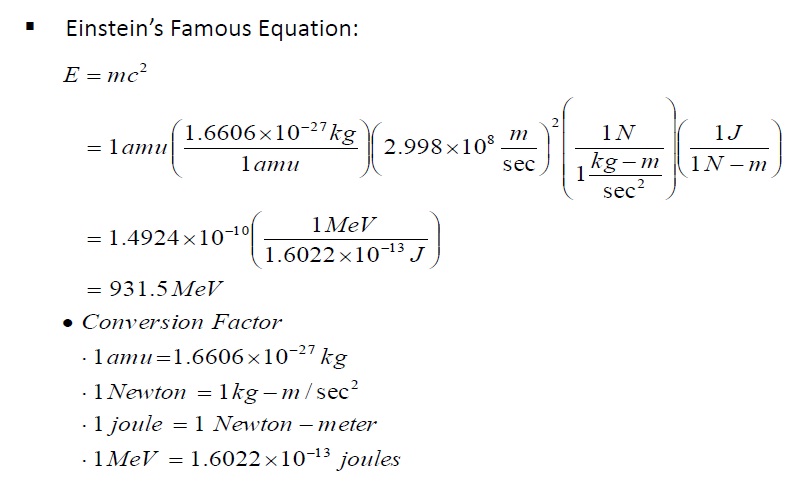
In nuclear physics, what is meant by the term 'mass defect', and what is its relation to nuclear energy? | Socratic