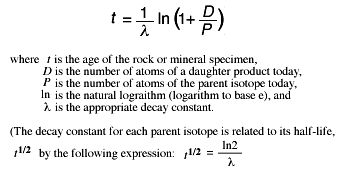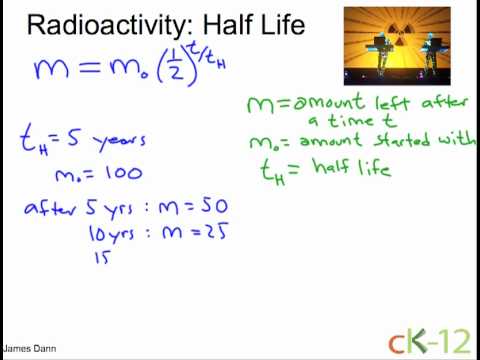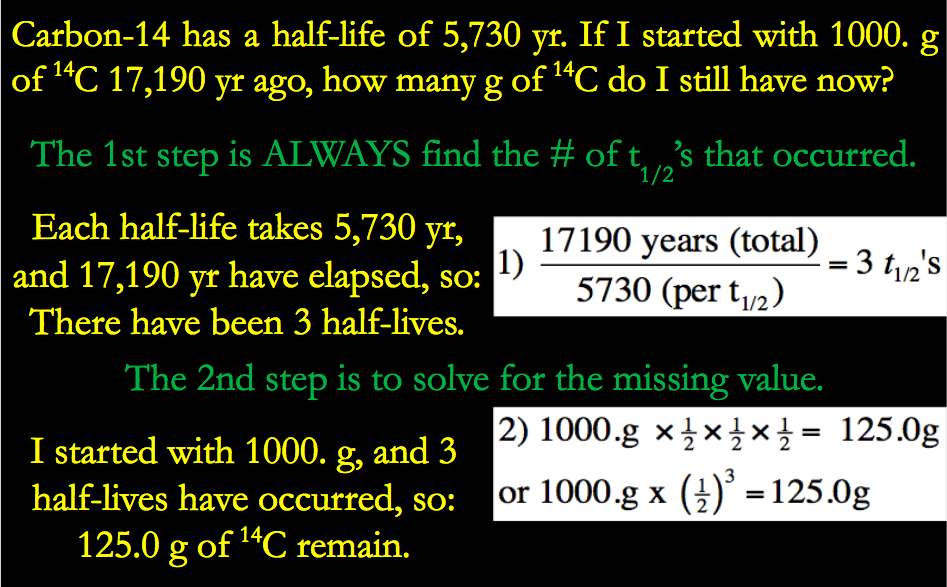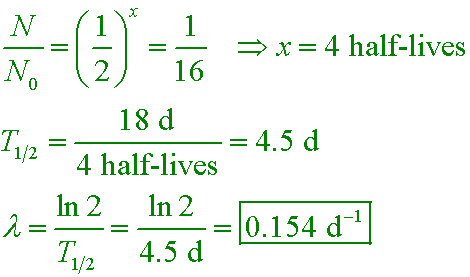How would you measure the half-life of a radioactive isotope, assuming the half life can be measured in minutes? - Quora
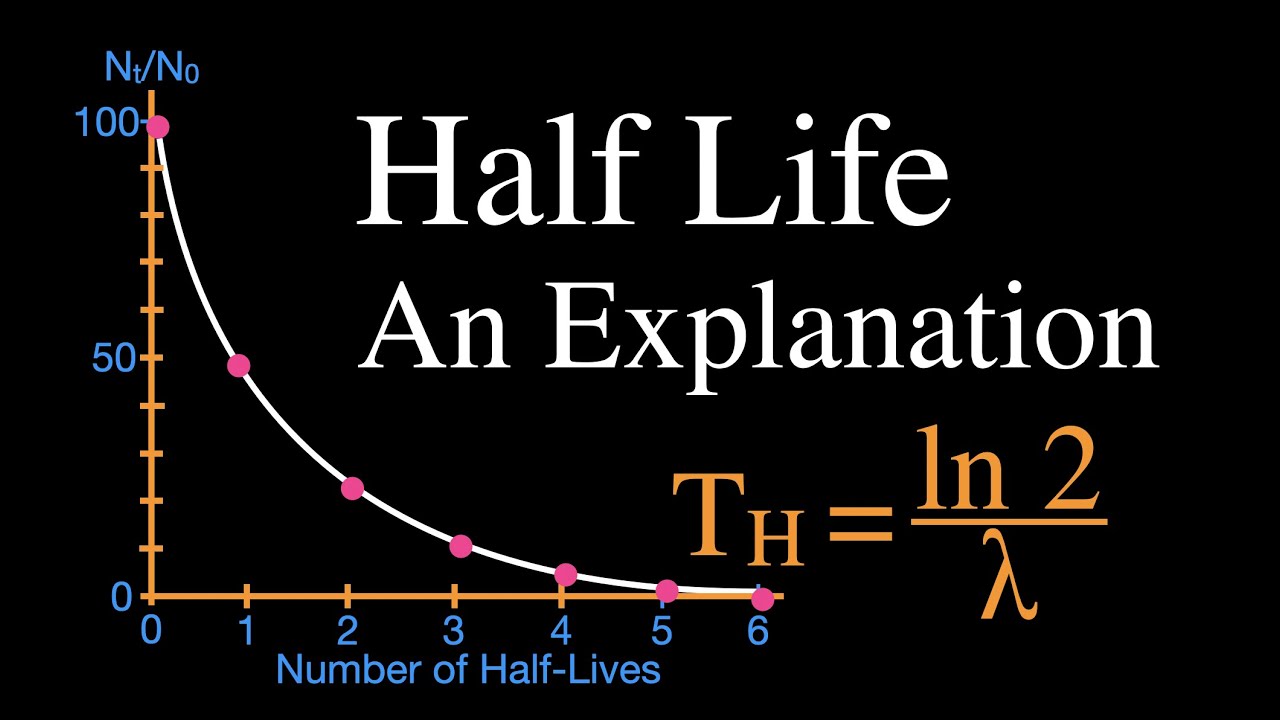
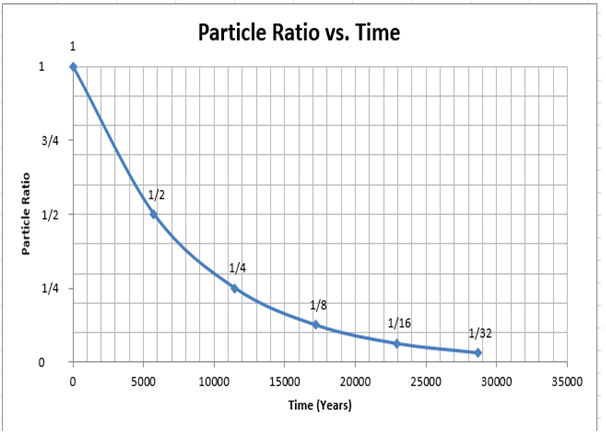
17.6: Radiocarbon Dating: Using Radioactivity to Measure the Age of Fossils and Other Artifacts - Chemistry LibreTexts

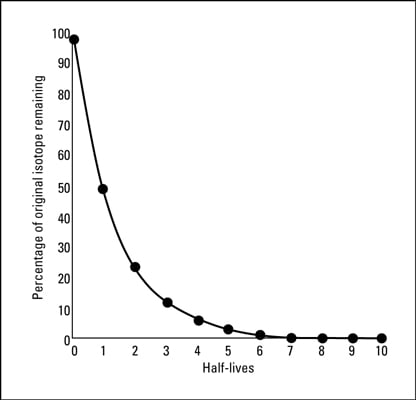
A radioactive isotope has a half - life of T years. How long will it take the activity to reduce to (a) 3.125% (b) 1% of its original value?
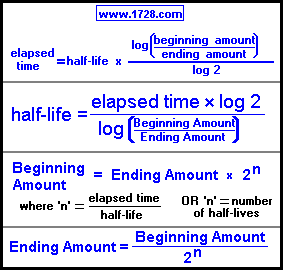
What is the half‑life of an isotope that decays to 12.5% of its original activity in 34.7 hours? - Quora