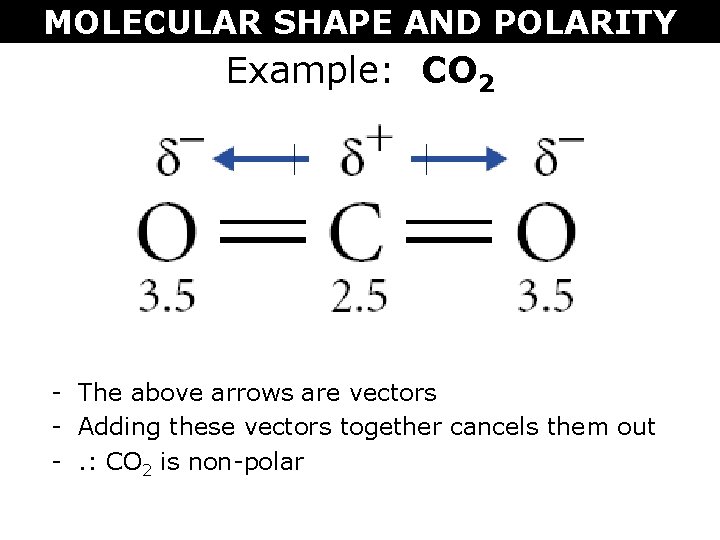
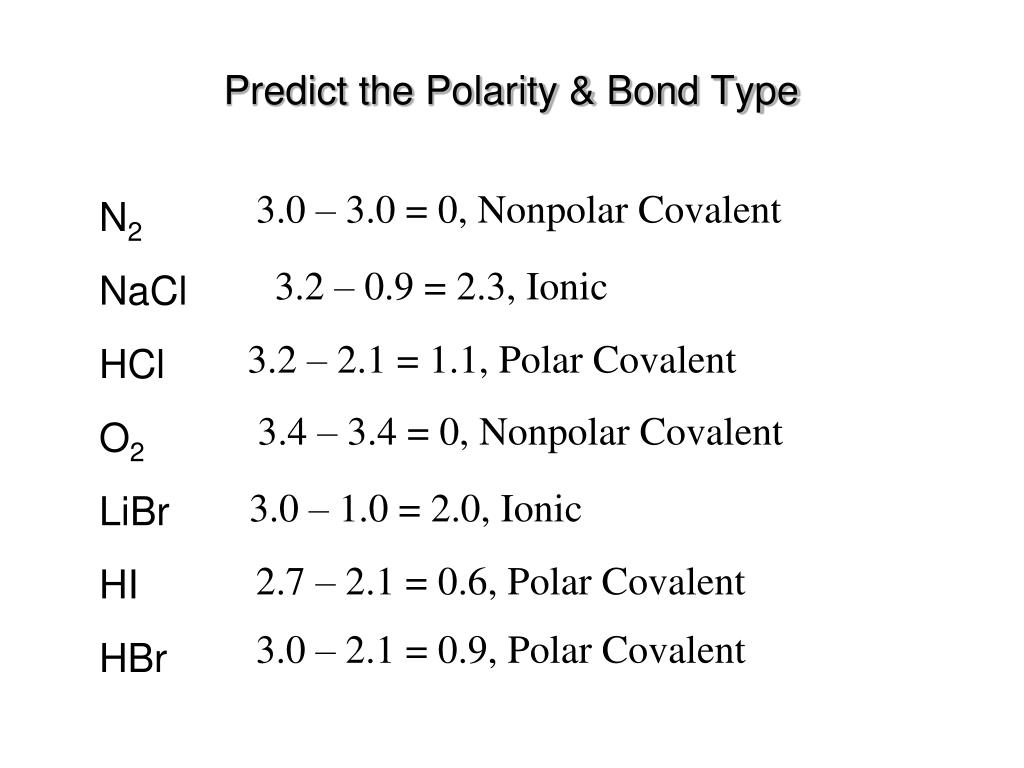
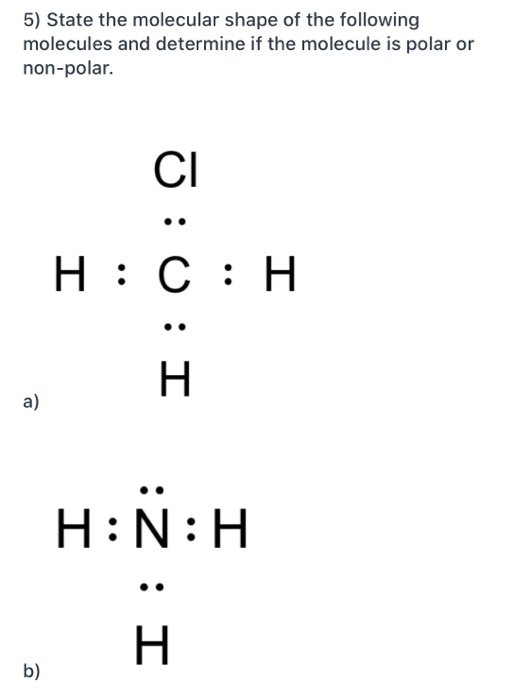
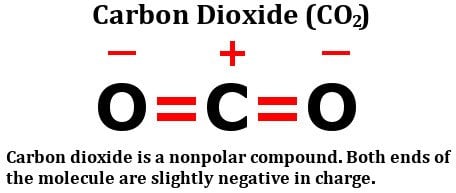1 Lewis Dot Formulas of Atoms Lewis dot formulas or Lewis dot representations are a convenient bookkeeping method for tracking valence electrons. Valence. - ppt download
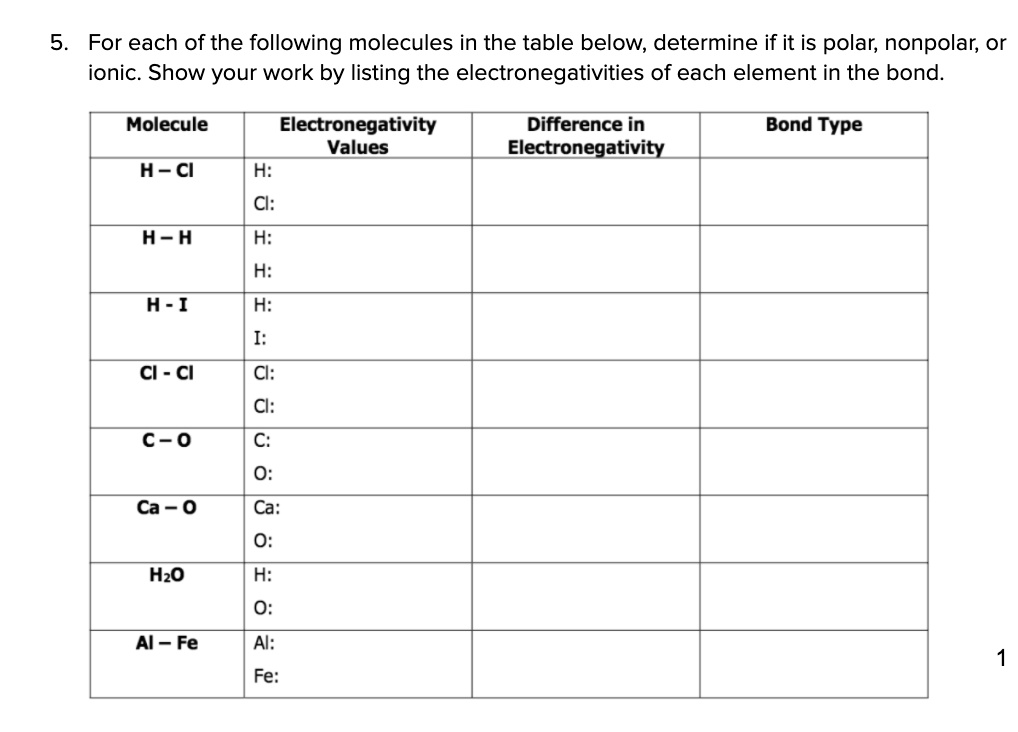
Chemistry 1110 Covalent Properties I. Assign electronegativity values to the following elements: N I S P H B O F Br C II. Tell
How is HI a polar molecule if its electronegative difference is 0.4? Doesn't it mean that it's a non-polar molecule? - Quora

REVIEW TIPS FOR BONDING EXAM OF MONDAY 3/7/16 1)IONIC CHARACTER IS A MEASURE OF POLARITY FOR POLAR COVALENT MOLECULES. H-Cl H-I H-F Electronagativity of. - ppt download

SOLVED:Activity 11.3 Determine which of the following molecules are polar; nonpolar; or ionic compound. 1 Hz 11. SO2 2 HI 12_ CaF 3 KCI 13. CaO 4_ CO 14_ CzHz 15. Clz






![No#1] Best Overview on Hydrogen Iodide (HI) - Science Education and Tutorials No#1] Best Overview on Hydrogen Iodide (HI) - Science Education and Tutorials](https://sciedutut.com/wp-content/uploads/2021/05/Hydrogen-iodide-transition-state-cover.png)