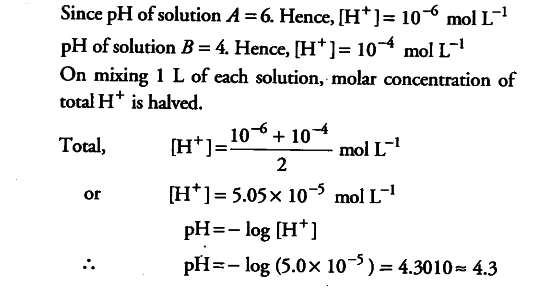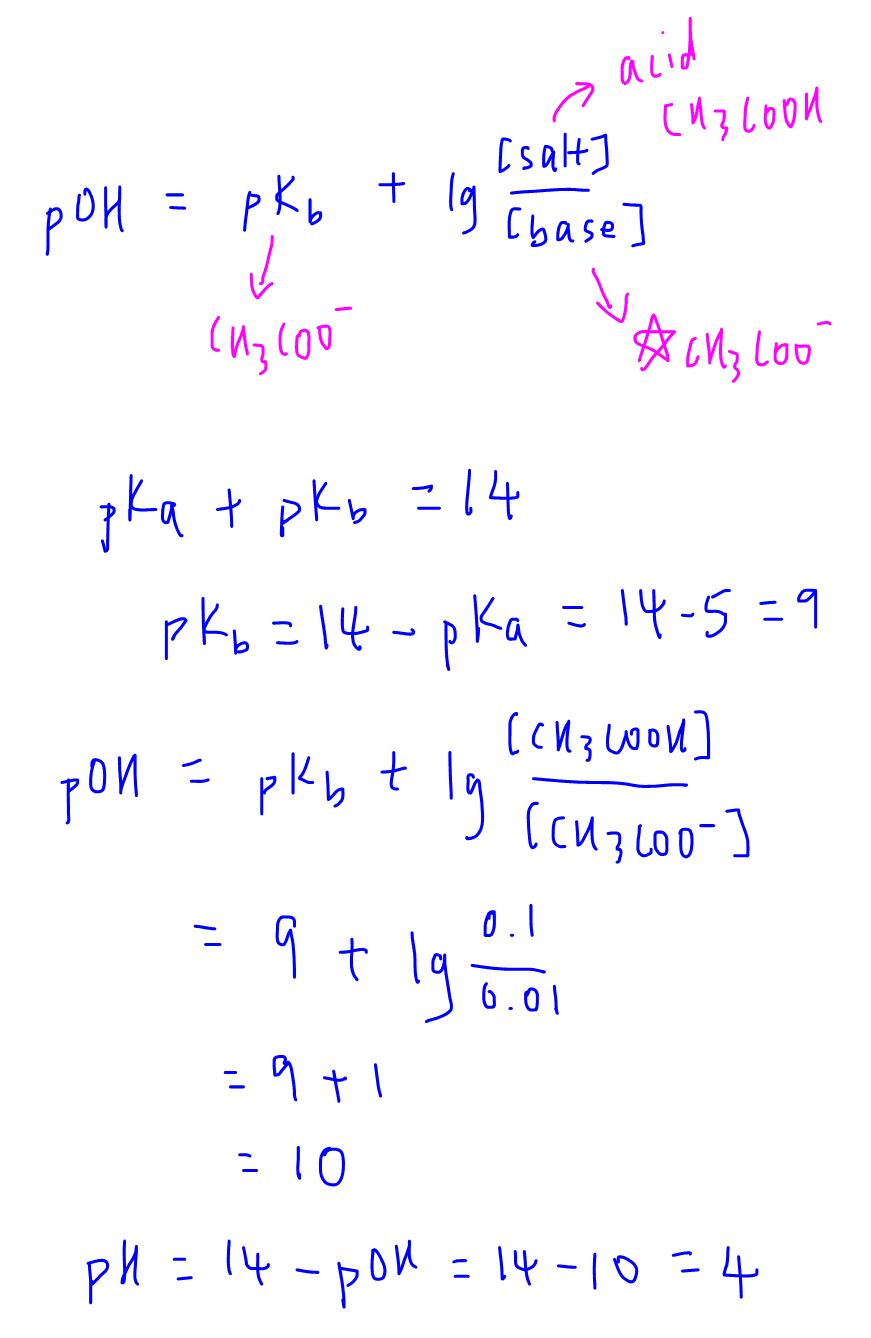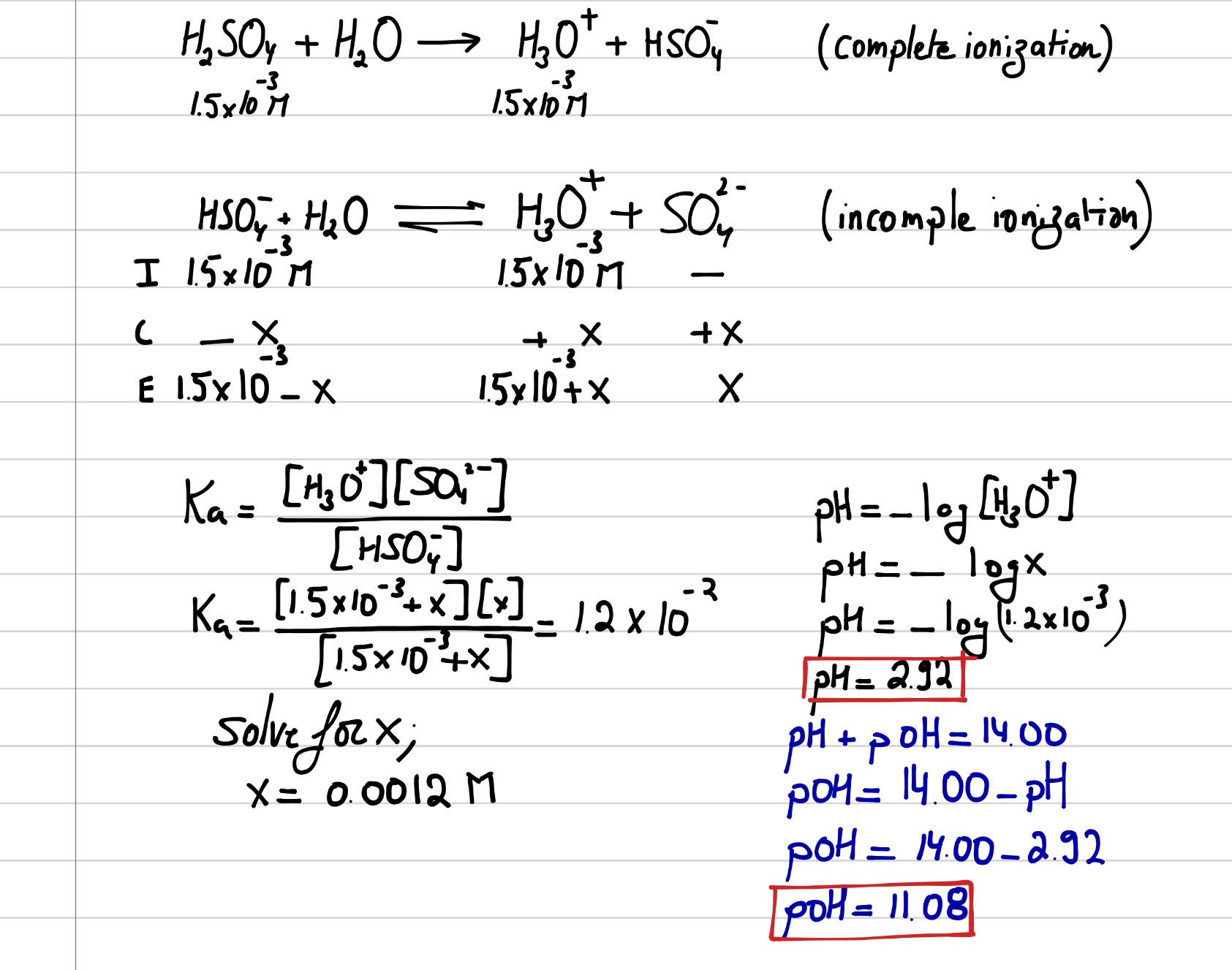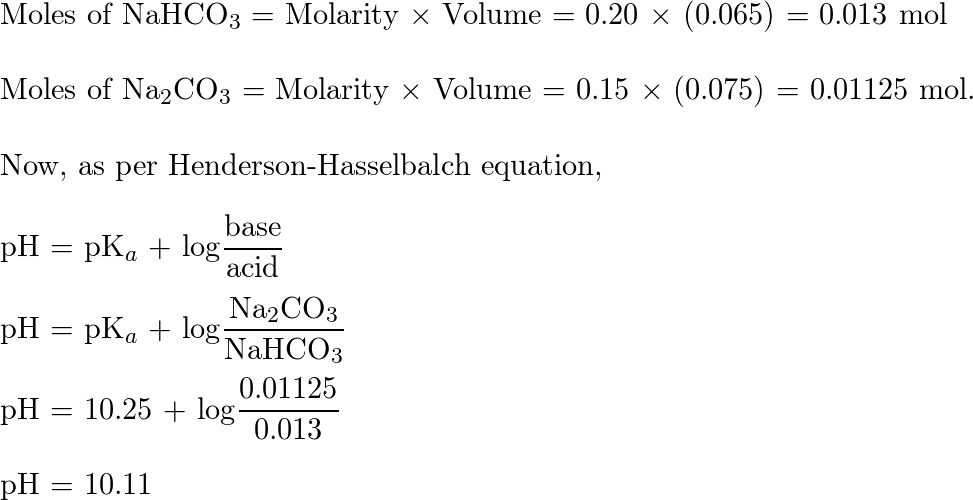The pH scale The pH scale is used to measure how acidic or basic a solution is. The lower the pH, the more acidic the solution. The higher the pH, the. -

SOLVED: Calculate the pH of a solution that contains 3.9 × 10-9 M H3O+ at 25°C. Calculate the pH of a solution that contains 3.9 × 10-9 M H3O+ at 25°C. 9.41 3.51 4.59 8.41 5.59