How to calculate the maximum mass of ammonia, NH3, that could be made from 42 tonnes of nitrogen and excess hydrogen - Quora

Question Video: Calculating the Equilibrium Constant for Concentration Given the Initial Amount of Each Reactant | Nagwa
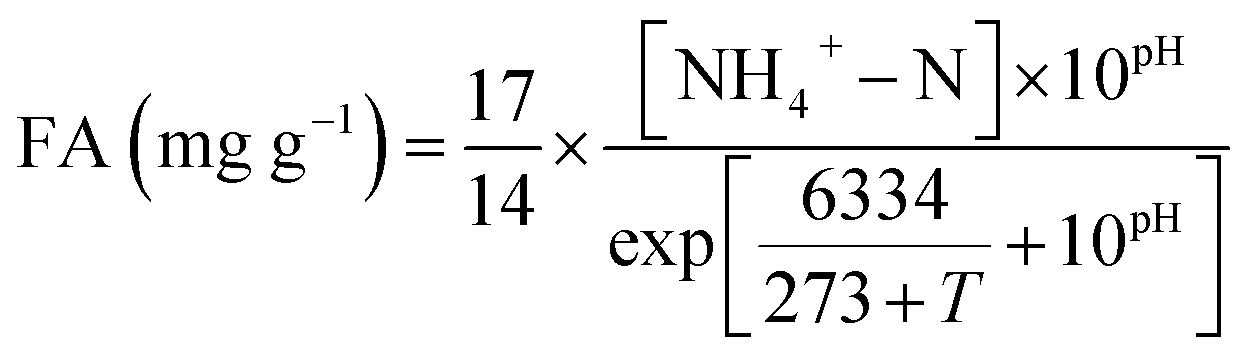
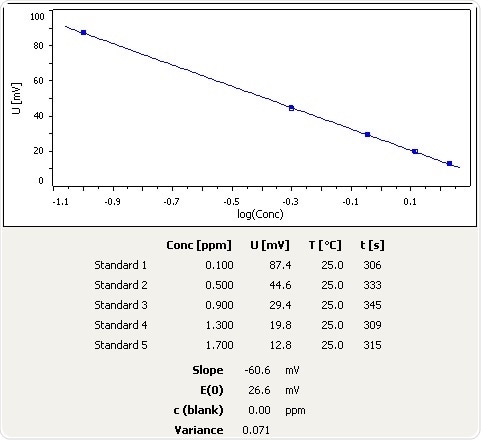
Characterizing the free ammonia exposure to the nutrients removal in activated sludge systems - RSC Advances (RSC Publishing) DOI:10.1039/C7RA10751J

stoichiometry - How to calculate the equilibrium constant for nitrogen, hydrogen, and ammonia? - Chemistry Stack Exchange
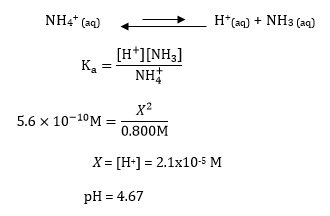
How to calculate your Un-ionized ammonia levels Information below collected from: Francis-Floyd, Ruth, Craig Watson, Denise Pett

SOLVED:The fertilizer, ammonium nitrate, is made by reacting ammonia with nitric acid. (Section 1.4) (a) Write a balanced equation, with state symbols, for the reaction of ammonia gas with nitric acid to

Reaction Mechanism and Kinetics for Ammonia Synthesis on the Fe(111) Surface | Journal of the American Chemical Society

Step-by-step guide to calculating unionized (toxic) ammonia. UIA of... | Download Scientific Diagram